Landmark WHO Approval for First Newborn-Specific Malaria Drug
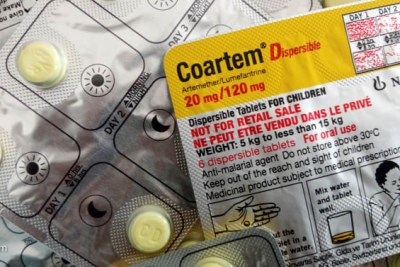
The World Health Organization (WHO) has approved the first malaria treatment developed exclusively for newborns and very young infants. The approval, announced on April 24, confirms that the treatment meets international standards for safety and effectiveness.
WHO Director-General Dr. Tedros Adhanom Ghebreyesus hailed the progress being made in the fight against the disease: "For centuries, malaria has stolen children from their parents, and health, wealth, and hope from communities". The WHO chief added that new vaccines, diagnostic tests, and next-generation mosquito nets are also helping to turn the tide.
The new artemether-lumefantrine formulation was developed through a partnership between Novartis and the Medicines for Malaria Venture (MMV), under the PAMAfrica consortium. In August 2025, the Africa Centres for Disease Control and Prevention (Africa CDC) praised the Swiss drug regulator's approval of the formulation while commending eight African countries for their participation in clinical trials.
InFocus
-
Despite preventable tools, cases are rising in Africa, prompting a global call to meet the UN's 2030 elimination goal with increased urgency. Malaria continues to claim the lives of thousands of people worldwide.
World Malaria Day is observed on April 25, highlighting the global fight to eradicate a mosquito-borne disease that is preventable but can be deadly. This year's theme is 'Driven to End Malaria: Now We Can. Now We Must', the global health community is being called
Read more »
-
Join us as we continue our in-depth series where we'll share the groundbreaking Malaria Consortium research about how malaria and Covid-19 intersect, and the high prevalence of this debilitating new condition called Long Covid.
This week allAfrica's Melody Chironda and Juanita Williams continue their conversation with Dr Jane
Read more »
-
The Africa Centres for Disease Control and Prevention (Africa CDC) has welcomed the approval of the first malaria treatment designed for newborns and infants under five kilograms, calling it a "major step forward".
The new artemether-lumefantrin formulation for children was developed through a partnership between Novartis and
Read more »
-
The country saw a decline in malaria infections for about a decade, with cases falling from nearly five million in 2016, to 430,000 in 2023 -- a 90% reduction. In 2024, however, malaria cases rose by more than 45%, prompting health authorities to reconsider an earlier decision to opt out of receiving malaria vaccines during the first phase of distribution by the World Health Organization and GAVI in 202
Read more »

New mothers in a Lilongwe hospital (file photo).