First-Ever Malaria Drug for Infants is Approved
The Africa Centres for Disease Control and Prevention (Africa CDC) has welcomed the approval of the first malaria treatment designed for newborns and infants under five kilograms, calling it a "major step forward".
The new artemether-lumefantrin formulation for children was developed through a partnership between Novartis and the Medicines for Malaria Venture (MMV), under the PAMAfrica consortium, and 8 African countries participated in clinical trials: Burkina Faso, Côte d'Ivoire, Kenya, Malawi, Mozambique, Nigeria, Tanzania, and Uganda.
Children under 5 were often given modified doses of medicines for older children, raising the risk of overdose and toxicity. This new formulation offers a safe, effective, and infant-friendly option, dissolvable in breast milk and with a sweet flavour to ease administration.
InFocus
-
The country saw a decline in malaria infections for about a decade, with cases falling from nearly five million in 2016, to 430,000 in 2023 -- a 90% reduction. In 2024, however, malaria cases rose by more than 45%, prompting health authorities to reconsider an earlier decision to opt out of receiving malaria vaccines during the first phase of distribution by the World Health Organization and GAVI in 202
Read more »
-
Ghana and Sierra Leone are using AI-powered drones to identify and eliminate mosquito breeding grounds in a bid to prevent malaria outbreaks.
The drones, developed by Japanese start-up SORA Technology, scan wetlands and riverbanks for stagnant water and deploy larvicide to stop mosquito eggs from hatching. Before the insects hatch, the drones deposit larvicide at the infestation point.
Africa accounts
Read more »
-
World Malaria Day, observed every year on April 25, shines a spotlight on the urgent need to sustain political commitment and investment in malaria prevention and control, especially in Africa, which bears 94% of global malaria cases and deaths.
Uganda has become the 19th African country to introduce the malaria vaccine
Read more »
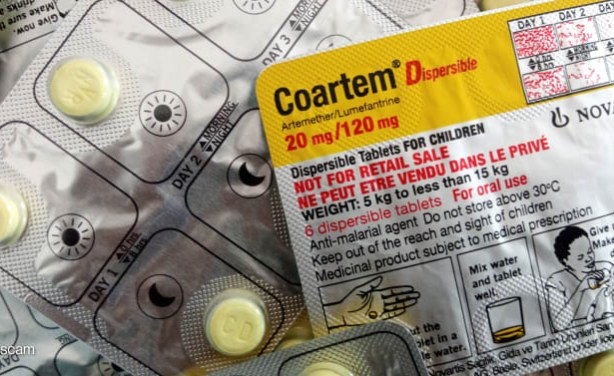
A new formulation by Swiss drug company Novartis AG for treating malaria in infants has been approved for use.









